What You Need to Know About Clinical Trials
When it comes to complex and aggressive conditions like pediatric retinoblastoma, clinical trials are a fundamental pillar in knowledge, research, and evolution of treatment. They’re an opportunity to learn more about these conditions, as well as how we can come one step closer to curing those who suffer from them.
Learn more about clinical trials and whether they may be right for your family.
What Is a Clinical Trial?
Clinical trials explore specific treatments, strategies, and devices for medical conditions to establish which options are safest and most effective.
In some cases, they can also help to determine the best treatment options for people in certain groups, like a certain stage or subtype of a disease.
Trials typically occur in the final stages of a long, careful research process. They follow strict scientific guidelines to help protect participating patients and ensure that the results are reliable.
How Clinical Trials Work
 Participating patients may get treatments or tests in a clinic, hospital, or doctor’s office. They’re all based on a master plan for every medical center participating in the study, called a protocol. The protocol outlines what exactly the trial will accomplish and why.
Participating patients may get treatments or tests in a clinic, hospital, or doctor’s office. They’re all based on a master plan for every medical center participating in the study, called a protocol. The protocol outlines what exactly the trial will accomplish and why.
Clinical Trial Phases
Trials that test medical devices and medicines will occur in phases, which help researchers to answer key questions along the way.
For example, a phase I clinical trial will explore safety and side effects in small groups of participants. Phase II will look at the efficacy of the treatments and continue safety reviews. Once these treatments have the green light, phase III will involve larger groups of participants to continue efficacy research, possible side effects, and compare the treatment with others on the market.
Benefits and Risks
Before deciding on participating in a clinical trial, it’s critical to weigh out the benefits and risks. It’s important to understand that while the treatments and tests being researched have shown promise to get to the clinical trial stage, they can still be harmful or ineffective.
Some of the benefits include:
- Having access to new, revolutionary treatments that aren’t yet available to the public
- Receiving expert medical care within a leading healthcare facility
- Helping others with similar conditions by contributing to the research needed to find a cure
Clinical trial risks include:
- The possibility of unpleasant, serious, or potentially life-threatening treatment side effects
- Some individuals’ conditions may not respond to treatment as well as others
- The trial may involve extensive travel time to the facility, extended treatment duration, or hospital stays
- Your health insurance plan may not cover the full costs of the study
Current Pediatric Retinoblastoma Trials
St. Jude Children’s Research Hospital has information on pediatric retinoblastoma clinical trials.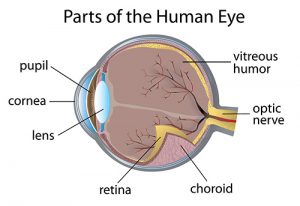
The iRET trial is for children 17 or younger who received a retinoblastoma diagnosis that came back or didn’t go away after their treatment. This study will examine the effects, impact, and potential cure rate of injecting carboplatin directly into the vitreous, of the gel that fills the majority of the eye.
The SJRET6 trial is for newly-diagnosed patients whose retinoblastoma has not spread beyond the eye, and who have not received previous radiation therapy or chemotherapy. This Phase II study will examine the clinical outcome of giving chemotherapy directly to the child’s eye with the first two cycles, or six weeks, of treatment.